The 4 sleep stages, any motor skill, and our brain — A deep dive
The 4 sleep stages, any motor skill, and our brain — A deep dive
Intro
The essence of being productive is to choose the right direction for our efforts. And of course, one has to make progress in that direction, the harder you work, the faster you move. That’s how the concepts of effectiveness (doing RIGHT things) and efficiency (doing things RIGHT) differ. As an essential part of being effective thus becomes — how can we do more in the same amount of time?
Again, there are multiple ways to approach this. One idea is to just work more “intensely”, another to find some tool or system to help us. But the most reliable and ironically most underrated way in our opinion is this — being better skilled. Wanna write blog posts quicker? Grammarly might help sure, but it’s nowhere close to the benefits which the ability to type 150 words per min will give in this context. Capische?
In a first, we bring to you a watered-down, more consumable version of a research paper. This time it's from the fascinating field of sleep science — a mix of biology, neuroscience, statistics, psychology, and more. The 2016 paper titled “Cued Reactivation of Motor Learning during Sleep Leads to Overnight Changes in Functional Brain Activity and Connectivity” is peer-reviewed, heavily cited, and published in a reputed journal.

The authors were James N. Cousins ,Wael El-Deredy,Laura M. Parkes,Nora Hennies,Penelope A. Lewis : a bunch of psychologists and Neuroscientists. You can find the original paper here. We will walk you through their research and then go on to bring it into perspective. We will finish by trying to see what we can learn or unlearn from this exercise on our journey to Consistent Productivity. Let’s go!
Context
The study was carried out at the Manchester Sleep Laboratory. It was supported by the UK’s Medical Research Council (MRC) and Biotechnology and Biological Sciences Research Council (BBSRC).
Twenty-five healthy participants aged 18–35 years volunteered. Prestudy questionnaires determined that participants had no history of psychiatric diseases, neurological, sleep, or motor disorders and kept a normal sleeping pattern in the week prior to the experiment.
Parts of the brain
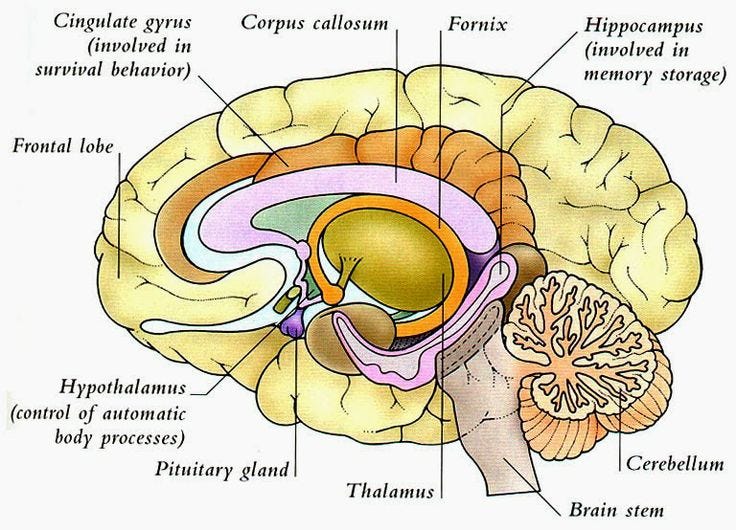
Before we begin, we need to get familiar with some of the key parts of the brain and their locations. The central nervous system (CNS) consists of the brain and spinal cord. The brain can be divided into the cerebrum, brainstem, and cerebellum. Refer to the images to understand the location of various sections.
The cerebrum is the largest division of the brain. It’s divided into 2 hemispheres, each of which is divided into 4 lobes. The cerebral cortex is its outermost layer.
The cerebellum (which is Latin for “little brain”) is a major structure of the hindbrain that is located near the brainstem. This part of the brain is responsible for coordinating voluntary movements. It is also responsible for a number of functions including motor skills such as balance, coordination, and posture.
Hippocampus is the part of the brain which is embedded deep in the temporal lobe of each cerebral cortex. It is an important part of the limbic system. The major role concerns the regulation of motivation, emotion, learning, and memory.
The Striatum is a nucleus (a cluster of neurons) in the subcortical basal ganglia of the forebrain. It coordinates multiple aspects of cognition, including both motor and action planning, decision-making, motivation, reinforcement, and reward perception. The striatum is made up of the caudate nucleus and the lentiform nucleus.
The caudate nucleus is a small yet very significant part of the brain that plays a combined role in monitoring motor functions, along with handling procedural learning, associative learning, and inhibitory actions (telling you when to not do something )
We might also mention other terns like SMC (Supplementary Motor Complex), right premotor cortex (PMC), etc. These are subsections of these main brain regions. As long as you have a rough idea of what part they belong too, you should be fine. Alright, here we go!
Sleep Stages
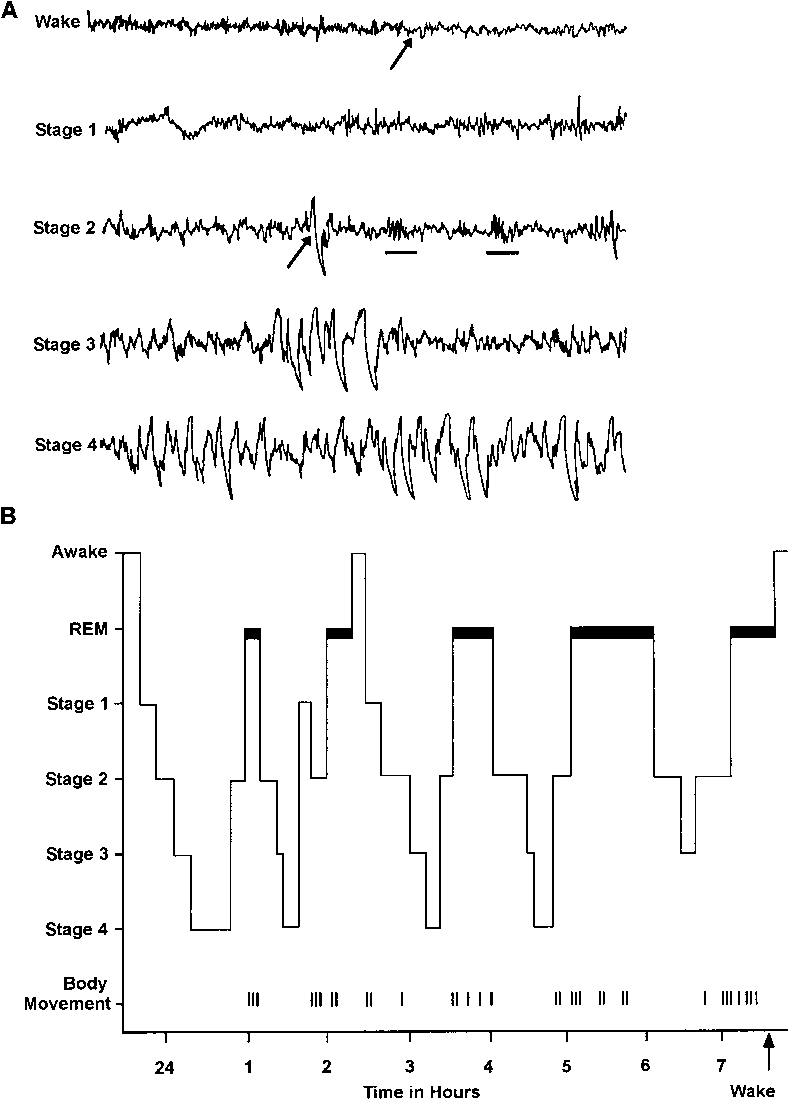
We ought to understand the 4 distinct stages of sleep as well. The diagram shows the sleep cycle in a full night for a normal adult on the top and the corresponding EEG patterns on the bottom. During sleep, the brain remains highly active, showing electrical activity in the form of ‘sharp-wave ripples’ in the hippocampus and large-amplitude slow oscillations in the cortex (the outer layer of the cerebrum), reflecting alternating periods of active and silent states of cortical (belonging to the cortex) neurons during deep sleep(or SWS).
These activities are measured by basically measuring voltage difference variations through electrodes affixed on our scalps. As we can see above, there are the aforementioned slow oscillations in stages 3 and 4 of the sleep cycle hence giving them a collective name of SWS. Stage 1 is aptly called the REM (rapid eye movement sleep) wherein our eyeballs move in fast random bursts while the eyelids are blissfully closed and the body is completely paralyzed while stage two is called the non-REM sleep.
The Task
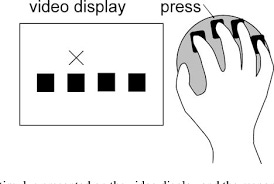
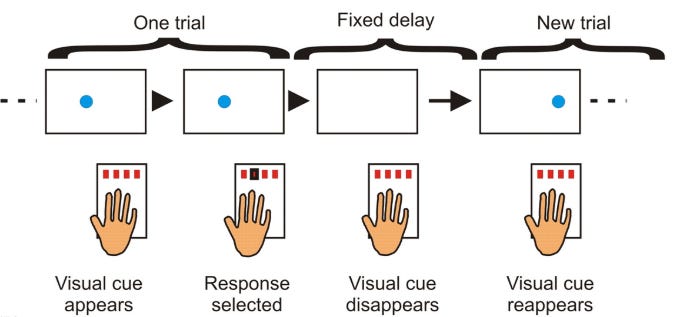
Participants participated in an adapted SRTT. Serial reaction time(SRT) is a commonly used parameter for measuring learning. In an SRT task (SRTT), participants are asked to repeatedly respond to a fixed set of stimuli (inputs — visual, auditory, etc) in which each cue signals that a particular response (i.e., button press) needs to be made. Probabilities are governing the transition between the cues, and thus participants can’t anticipate what is coming next.
There are fixed patterns (sequences) that come up. But they are randomly introduced and interspersed with blocks that have no fixed sequence as such. You are expected to press the correct button as quickly as possible; i.e- reduce your response time, while pressing the correct button as many times as possible. Hence, there is a trade-off between speed and accuracy.
The task for this experiment contained pseudorandomly interleaved blocks of two 12-item sequences, A (1–2–1–4–2–3–4–1–3–2–4–3) and B (2–4–3–2–3–1–4–2–3–1–4–1), with no runs of more than two blocks of the same sequence.
Trials contained an auditory tone and visual cue in one of four spatial locations, corresponding to a four-button box used with all fingers of the left hand. “A” or “B” appeared centrally on the screen to indicate the sequence.
Cueing each sequence on-screen was accompanied by pure tones, four high-pitched tones were used for one sequence (fifth octave (Pa); A/B/C/D), and four lower-pitched tones were used for the other (fourth octave (Ma); C/D/E/F).
The Experiment Design
Sequences A and B were equally divided across cueing conditions so that half were cued during sleep with sequence A and a half with sequence B.
Participants were invited to sleep overnight in the Neuroscience and Psychology of Sleep (NaPS) Laboratory at the University of Manchester, where they were monitored with Polysomnography. (Polysomnography records your brain waves, the oxygen level in your blood, heart rate and breathing, as well as eye and leg movements during the study). Electrodes were affixed at standard locations for EEG monitoring of brain activity during sleep.
During periods of SWS (deep Non-REMsleep), one sequence’s tones were replayed just loud enough to be audible above the brown noise (at approximately 48 dB). They were replayed in the same order as learning and at a speed similar to their average performance pre-sleep. It was done in blocks of 2 min replay (CUE), followed by 2 min silence (NO-CUE).
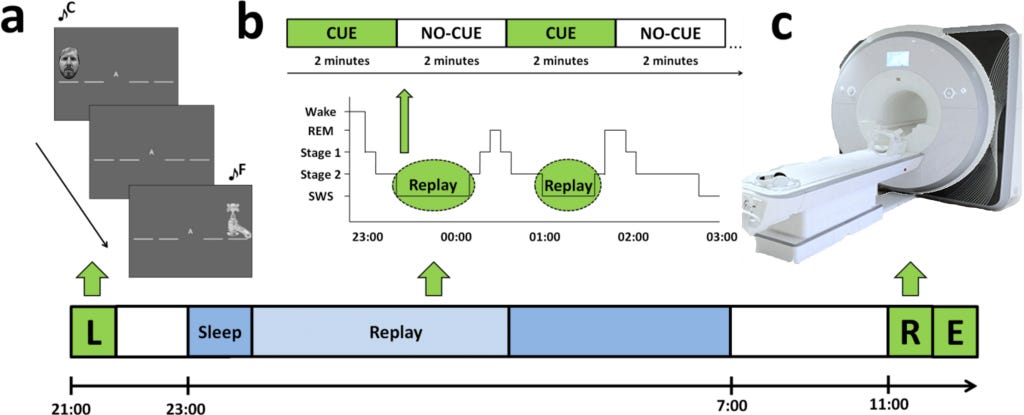
Order of learning (i.e., whether participants began a session with sequence A or B), replay, and retest was counterbalanced across participants. The volunteers were retested the next morning by cueing a modified version of the SRTT. The researchers measured subsequent differences in functional activity and connectivity during retest using functional magnetic resonance imaging (fMRI). Lastly, free recall was measured outside the scanner with participants marking sequence order on paper. The Stanford Sleepiness Scale assessed alertness prior to learning and retest sessions.
The point of such research
The role of sleep in memory consolidation is demonstrated by improved performance and neural plasticity underlying this improvement. Neural plasticity is the ability of the central nervous system to adapt to changes in the environment through growth and reorganization of the neurons.
The memory undergoes “offline” processing after a motor skill is learned so that improvement occurs even without further practice. It’s still uncertain how sleep is able to do so, and whether this process is affected by various stages of sleep.
To test this in humans, a replay of specific memories is cued by the presentation of a sound that was being played during learning/ performing the task. This is termed as Targeted Memory Reactivation (TMR).

Sleep-dependent consolidation is manipulated through intentionally biasing the replay of specific memories in sleep, but the underlying neural basis of these altered memories remains unclear.
The observations from such research give further insight into sleep’s unique role in memory consolidation which is shown by offline skill enhancement. This depends on the reactivation of specific memories, and the associated changes in neural activity may rely upon processing that occurs during different stages of sleep.
Background
Memory consolidation begins as soon as the new information is encoded in our brain. It is a process of stabilizing, strengthening, and reorganizing fragile memories. Learning a new motor skill, for example, requires episodes of repeated practice, and is also supported by offline consolidation periods where stabilization and gains in performance are observed.
These performance improvements are a result of plastic changes within key motor memory networks over a period of time. Sleep provides optimal conditions for offline processing to occur. This statement is suggested by several studies contrasting sleep and wake consolidation periods.
The reason for such plasticity during sleep and the associated performance gains has been hypothesized to be the spontaneous reactivation of cerebral activity. This observation of “memory replay” has been made in multiple brain regions during sleep in rodents and humans.
Overnight procedural memory consolidation is linked to enhanced functional activation within the striatum, hippocampus, cerebellum, and motor cortical regions, as well as strengthening of ceratin connections between such parts.

The reason it’s hypothesized that TMR would modulate changes in functional activity and connectivity is the reactivation, which is thought to drive the plasticity connected with sleep. The jury was still out on the role of REM sleep in procedural memory reactivation and consolidation, as of the writing of this paper.
The objective of this paper
No evidence has been found to link REM with memory reactivation. The major objective of the paper was to explore what role do both REM and NREM play in memory consolidation by examining how brain functions after TMR are influenced by individual differences in REM and SWS.
To reiterate, the test was done by cueing a modified version of the serial reaction time task (SRTT) during SWS. One sequence (cued) was reactivated during nocturnal SWS by replaying the associated tones in sequence, while the other sequence (uncued) was not. Brain activity and behavioral measures of speed and accuracy were compared while performing cued and uncued sequences at postsleep retest.
Results — Behavioural Analysis — Reaction/Response Times (RT)
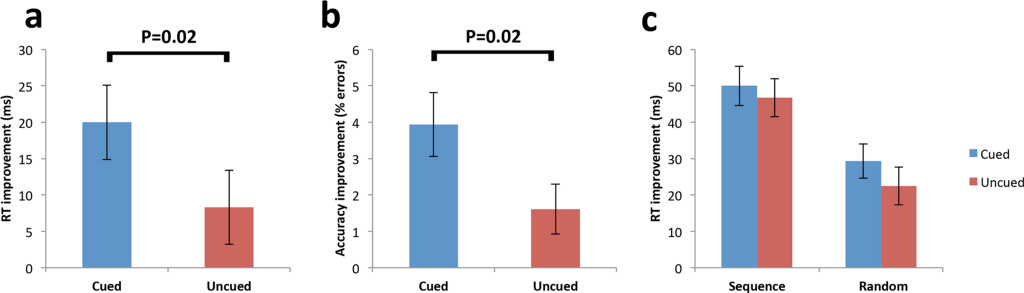
Firstly, it was confirmed that sequence learning (one task is learned before the next) occurred prior to sleep, by showing that RTs were significantly faster for sequence trials compared to random trials (tasks are learned without any structure) for both cued and uncued sequences.
Therefore, any post-sleep differences would have to be attributed to the TMR of the cued sequence (since all other external factors have been suitably negated).
Next, they explored early improvement in RTs due to TMR effects by subtracting initial blocks (a parameter=number of correct responses * reaction time) of sequence retest (post-sleep) from the final blocks of sequence learning (pre-sleep performance), providing a measure of “early sequence improvement.” Here, they found improvement for the cued sequence was significantly greater than the uncued.
Next, they compared cued and uncued sequence performance during the last blocks of the retest (late sequence improvement) and found that the advantage for the cued sequence was no longer present (residual block size is nearly 0). The lack of an effect at later blocks most likely portrays the cued sequence approaching an upper limit, prior to the uncued sequence.
They suggest that this early improvement reflects an improvement in sequence learning rather than visuomotor mapping (=improvement in specific hand-eye co-ordination for the task).
Results — Behavioural Analysis — Error rates
Performance of the SRTT can be seen as a trade-off between accuracy and speed (maximizing for one will lead to poor performance in the other).
Therefore, they analyzed errors across the same test blocks (same set of Q/As) as the RT analysis to ensure their cueing effects for RT were not due to an alteration in the speed-accuracy trade-off.
Random trials showed no difference between cued and uncued prior to sleep. After sleep, there was a significant reduction in the error rate for the cued relative to the uncued sequence at early blocks.
Significant RT enhancement for the cued sequence across early blocks represents a gain in speed, rather than being the result of a shift in the speed-accuracy trade-off.
Results — Functional Imaging Analysis
This is done by measuring the change in the different parts of the brain for cued vs uncued sequences postsleep. We already know many details about what role various brain parts play. From the imaging analysis (done using fMRI), researches try to see if they can find any meaningful correlations.
They investigated what factors were related to the cueing effect by running the same cued vs. uncued contrast with five regressors : Duration (mins) of SWS (SWStime), of REM (REMtime) and that of stage 2 (S2time), the number of replayed sequences (replays) and the procedural cueing effect.
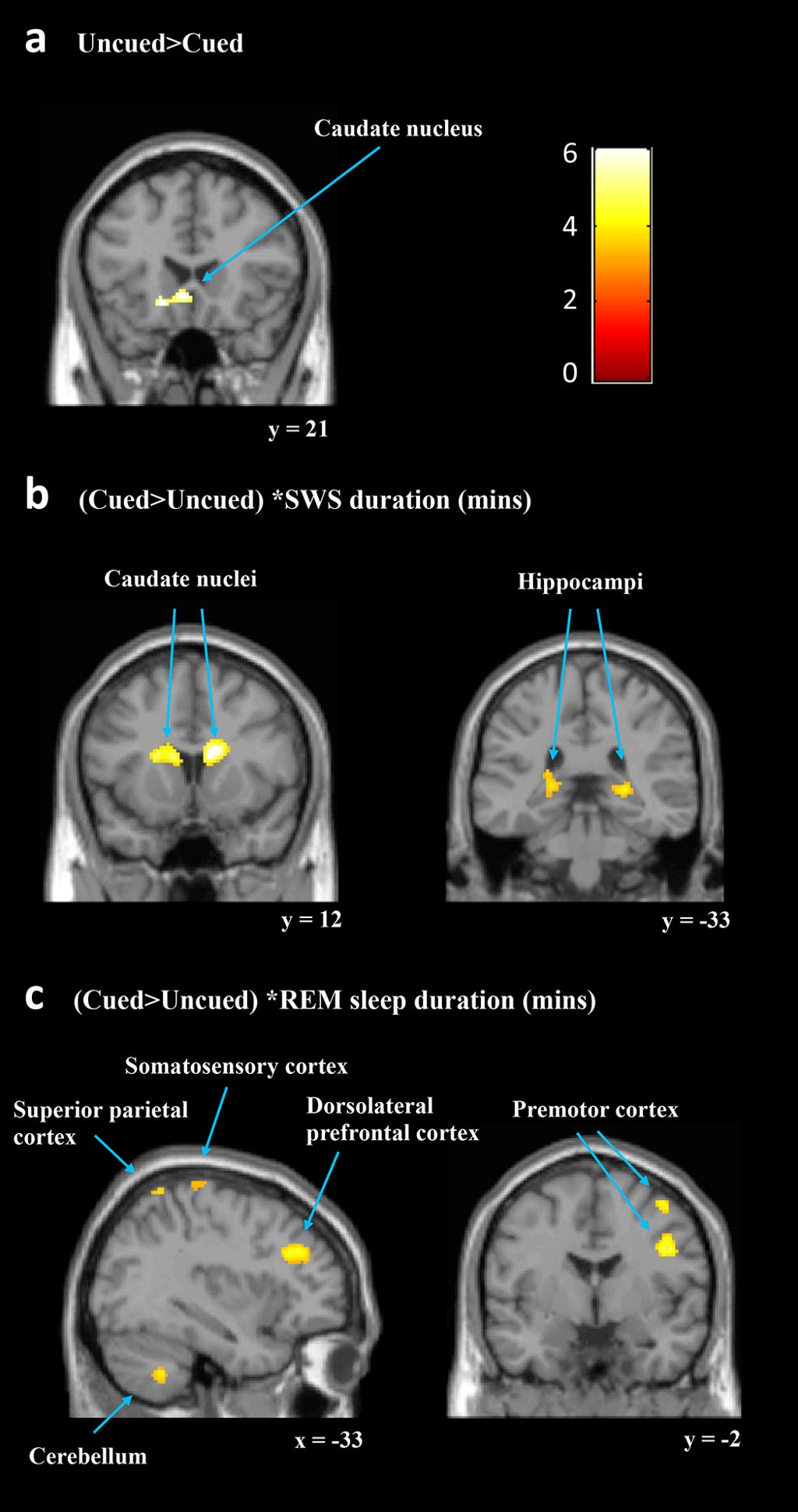
In sum, a longer duration of SWS was associated with increased responses to the cued sequence within predicted subcortical regions that are critical to procedural learning (caudate and hippocampus) while other key cortical motor regions showed decreased responses (SMC and FEF).
They next considered TMR effects with REMtime as the regressor of interest [(cued > uncued) * REMtime]. This showed significant clusters of increased activation for the cued sequence in a number of motor regions, including the bilateral cerebellum, right premotor cortex (PMC), and left SMC.
Interestingly, this regressor also revealed decreased activity for the cued sequence in a right caudate cluster, which overlapped with the right caudate cluster that showed increased activity when considering SWS. Thus, REM and SWS were associated with changes in activity after cueing in separable motor learning regions and also had opposite relationships with activity levels in the right caudate. This was an important observation.
The replays regressor showed the reverse pattern to the SWS regressor. Procedural cueing effect as a regressor [(cued > uncued) * procedural cueing effect] did not identify increases in any of the predicted regions, and there were no significant decreases.
Results — Functional Connectivity Analysis
After identifying localized activation differences associated with TMR during SWS, they sought to examine the functional connectivity of task-related regions that showed sensitivity to TMR. What this basically means is measuring how and if connections between various parts of the brain have changed.
According to Wikipedia, Psychophysiological interaction (PPI) is a brain connectivity analysis method for functional brain imaging data, mainly fMRI. It estimates context-dependent changes in effective connectivity (coupling) between brain regions. Thus, PPI analysis identifies brain regions whose activity depends on an interaction between psychological context (the task) and physiological state (the time course of brain activity) of the seed region.
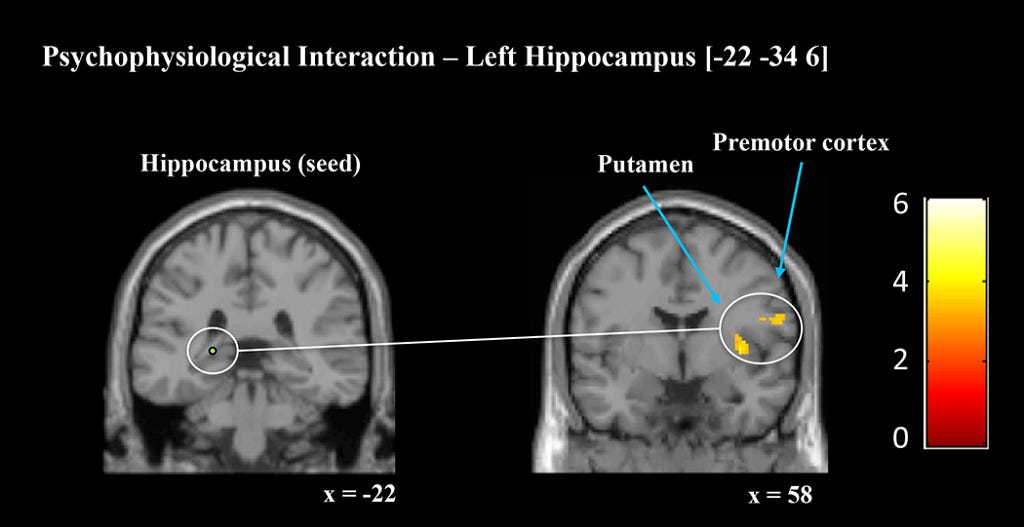
The connectivity strengths were measured by 4 PPI analyses seeded in right and left hippocampus and right and left caudate nucleus. Each analysis explored how connectivity from the seed region to the whole brain differed between cued and uncued sequences.
Crucially, both hippocampal seeds showed enhanced connectivity with key motor regions during cued relative to uncued sequence performance. Additionally, both the left and right caudate seed showed enhanced connectivity.
To summarise, TMR was associated with increased subsequent connectivity and activation within regions associated with procedural learning. This altered pattern of brain activity may underpin the behavioral enhancements that were observed during the retests postsleep.
Observations, hypotheses, and discussion
In their research, it was shown that targeted reactivation of a procedural memory alters functional activity and connectivity of motor memory networks in the human brain.
These results combined to support the distinct contributions of different sleep stages to consolidation. It is known that SWS facilitates consolidation in the striatum and hippocampus which notably support sequence learning. REM sleep contributes to motor learning by facilitating consolidation in cortical and cerebral networks.

A particularly significant finding is the association of REM with changes in functional activation, even though TMR occurred during preceding periods of SWS. This suggests a link between NREM memory reactivation and the processing that occurs in subsequent REM sleep. A controversial aspect creeps in regard to the role of REM in the reactivation and consolidation of procedural memories.
The ‘Sequential Hypothesis’ from the researchers proposes that REM strengthens and stabilizes the processes which began during preceding SWS periods which enabled synaptic consolidation to stabilize memories after being reorganized during SWS. The findings aid this model. The findings also suggest that reactivated procedural memories may undergo processing in separable REM and SWS networks.
The enhanced response speed for the cued sequence occurred alongside increased caudate and hippocampal responses associated with time spent in SWS. The increase in hippocampal response for the cued sequence is important for several reasons. Such enhanced activity is associated with overnight changes in motor sequence performance, and the hippocampus is hypothesized to be critical for engaging sleep-dependent sequence consolidation.
While the SWS-associated functional increases for the cued memory occurred in subcortical regions known for their roles in many types of learning, REM-associated increases related to cortical and cerebellar regions that are for the most part motor learning specifically. These include increased activity in the bilateral cerebellum, right ventral PMC, and left SMC.

Procedural consolidation has been earlier correlated with the duration of Stage 2. Several proposals have also been made regarding the optimal neurophysiological conditions for systems consolidation during stage 2 reactivation. This led them to examine NREM sleep duration as a whole clubbing the SWS and the Stage 2 duration. The NREM covariate provided virtually identical results to the SWS covariate, indicating a strong influence of SWS.
Conclusion
From this research, it can be inferred that TMR of a procedural memory alters functional activity and connectivity changes in striatum and hippocampus. The behavioural effects associated with TMR can be explained with this altered function. The study also hints at both REM sleep, as well as SWS being important for neural changes that support enhanced postsleep performance of a procedural skill.
The findings clearly support our understanding of sleep’s unique role in memory consolidation by showing that offline skill enhancement depends on the reactivation of specific memory traces. The findings also suggest associated changes in neural activity rely upon processing which may take place across several subsequent sleep stages.
Food for thought
Clearly, messing with our sleep is a bad idea. But the contrarian question is — can we use research about our brain to make us more effective (productive)? It’s common knowledge among Neuroscientists that the answer is an overwhelming yes. We are wired in a certain way and evolved in certain environments to fulfill certain functions.

So this paper and many more like it in this space seem to suggest this — Wanna learn a new skill? Touch Typing? Guitar? Snooker? Giving it more “practice” is not the only answer. You can choose to DIVIDE that practice more consistently across multiple days for better results.
3 days with 20 min of practice are better than 1 day of an hour of practice with 2 “blank” days following it. Also, our brain has a “recency bias” wherein memories or circuits that were last updated/activated tend to gain a priority when the brain shifts into “random access mode”.
It’s no surprise that people report having dreams about stuff they were involved in right before sleep. Perhaps we can all schedule our last practice session for the day right before shut-eye to give our brain the highest chance to pick that specific skill up for consolidation — on priority.
We are still only scratching the surface in our understanding of the brain and also in the role that sleep plays. But we know enough to start. Enough to start hacking our brain to our advantage, to start learning how it functions to fine-tune our performance, to start consciously evolving into StrangerSapiens.
The 4 sleep stages, any motor skill, and our brain — A deep dive was originally published in StrangerSapiens on Medium, where people are continuing the conversation by highlighting and responding to this story.